|
Case Report
Successful treatment of multidrug-resistant Acinetobacter baumannii ventriculitis with continuous ventricular irrigation combined with intravenous tigecycline: A case report
1 Department of Neurosurgery, Shenzhen Nanshan People’s Hospital (Affiliated Nanshan Hospital of Shenzhen University), Shenzhen, China
2 Department of Critical Care Medicine, Affiliated Hospital of Guangdong Medical University, Zhanjiang, China
Address correspondence to:
Guoqiang Qin
Department of Neurosurgery, Shenzhen Nanshan People’s Hospital (Affiliated Nanshan Hospital of Shenzhen University), Shenzhen,
China
Message to Corresponding Author
Article ID: 100019Z16YC2026
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Chen Y, Feng Y, Huang X, Qin G. Successful treatment of multidrug-resistant Acinetobacter baumannii ventriculitis with continuous ventricular irrigation combined with intravenous tigecycline: A case report. J Case Rep Images Infect Dis 2026;9(1):1–8.ABSTRACT
Introduction: To report a case in which multidrug-resistant Acinetobacter baumannii (MDRAB) ventriculitis was successfully treated using continuous ventricular lavage combined with intravenous tigecycline, providing a reference for the clinical management of such complex intracranial infections. Methods: This was a retrospective analysis of the diagnosis and treatment process for a patient with extensive MDRAB ventriculitis following moyamoya disease surgery. The treatment regimens included (1) intravenous combination therapy with high-dose tigecycline, cefoperazone/sulbactam, and meropenem; (2) neuroendoscopic ventriculomyelocele evacuation; and (3) continuous ventriculostomy irrigation with tigecycline solution via a ventriculostomy tube.
Case Report: A 50-year-old female patient was transferred to the Neurosurgery Intensive Care Unit at Huazhong University of Science and Technology Union Shenzhen Hospital. After 62 days of intravenous tigecycline and 19 days of intraventricular tigecycline irrigation combined with neurosurgical intervention, the patient’s cerebrospinal fluid (CSF) white blood cell count decreased from 27,773 × 106/L to normal levels, and the intracranial infection was controlled. Transient coagulation disorders and elevated liver enzymes occurred during treatment but improved after symptomatic management. The patient ultimately improved and was discharged. At the 1-year post-operative follow-up, no recurrence of infection was observed.
Conclusion: For extensively drug-resistant AB ventriculitis in which first-line agents are unavailable because of drug availability or blood–brain barrier limitations, continuous ventricular lavage combined with intravenous tigecycline represents an effective salvage strategy. The success of this approach relies on multidisciplinary collaboration, thorough pharmacovigilance, and individualized neurosurgical management.
Keywords: Acinetobacter baumannii, Continuous ventricular irrigation, Multidrug resistance, Tigecycline
Introduction
Acinetobacter baumannii (AB) is a common causative agent of opportunistic nosocomial infections; the main sites of infection include the lungs, brain, urinary system, and blood system. Intracranial infections account for approximately 3.6% to 11.2% of AB infections [1]. In recent years, with the increased use of antibiotics, the incidence of multidrug resistance has reached 79.9% worldwide and up to 100% in some countries [2]. Patients with AB infections have poor outcomes, with a mortality rate of 38.4% in China and a global average mortality rate as high as 42.6%, making AB infections a major contributor to the global health burden. Because of the blood-brain barrier, many antimicrobial agents cannot penetrate the cerebrospinal fluid circulation, and even sensitive drugs exhibit low permeability. Consequently, clinical treatment of intracranial infections caused by multidrug-resistant Acinetobacter baumannii (MDRAB) poses significant challenges. Tigecycline, the first novel glycylcycline-class antibiotic, has not been approved by the U.S. Food and Drug Administration (FDA) for intracranial infections because of its difficulty in crossing the blood-brain barrier. However, in vitro drug sensitization and clinical trials have shown that tigecycline has a potent antibacterial effect on a variety of drug-resistant bacteria [3],[4]. In patients with purulent ventriculitis, pathogenic bacteria often accumulate in large quantities within the ventricles, skull base, and subarachnoid space. Continuous ventricular irrigation (CVI) of antibiotic fluids through a tube can eliminate bacterial colonization, and flocculation with a rinsing fluid can significantly reduce the concentration of bacteria in the cerebrospinal fluid (CSF) and limit the growth of bacteria in the flow state. Direct intraventricular administration allows drugs to enter the CSF pool and diffuse to the brain surface, achieving effective therapeutic concentrations and enhancing antimicrobial efficacy [5]. Therefore, combined intravenous (IV) and intraventricular administration may be an option to ensure CSF sterilization while minimizing adverse systemic reactions [5]. Currently, most reports of the use of tigecycline for intracranial AB infections are case series. Table 1 summarizes the patient demographics, dosing regimens, and clinical outcomes from the selected studies. In this study, we report the successful treatment of MDRAB-induced ventriculitis with CVI combined with IV tigecycline, with the aim of providing a reference for clinical practice.
Case Report
Basic Information and Initial Treatment
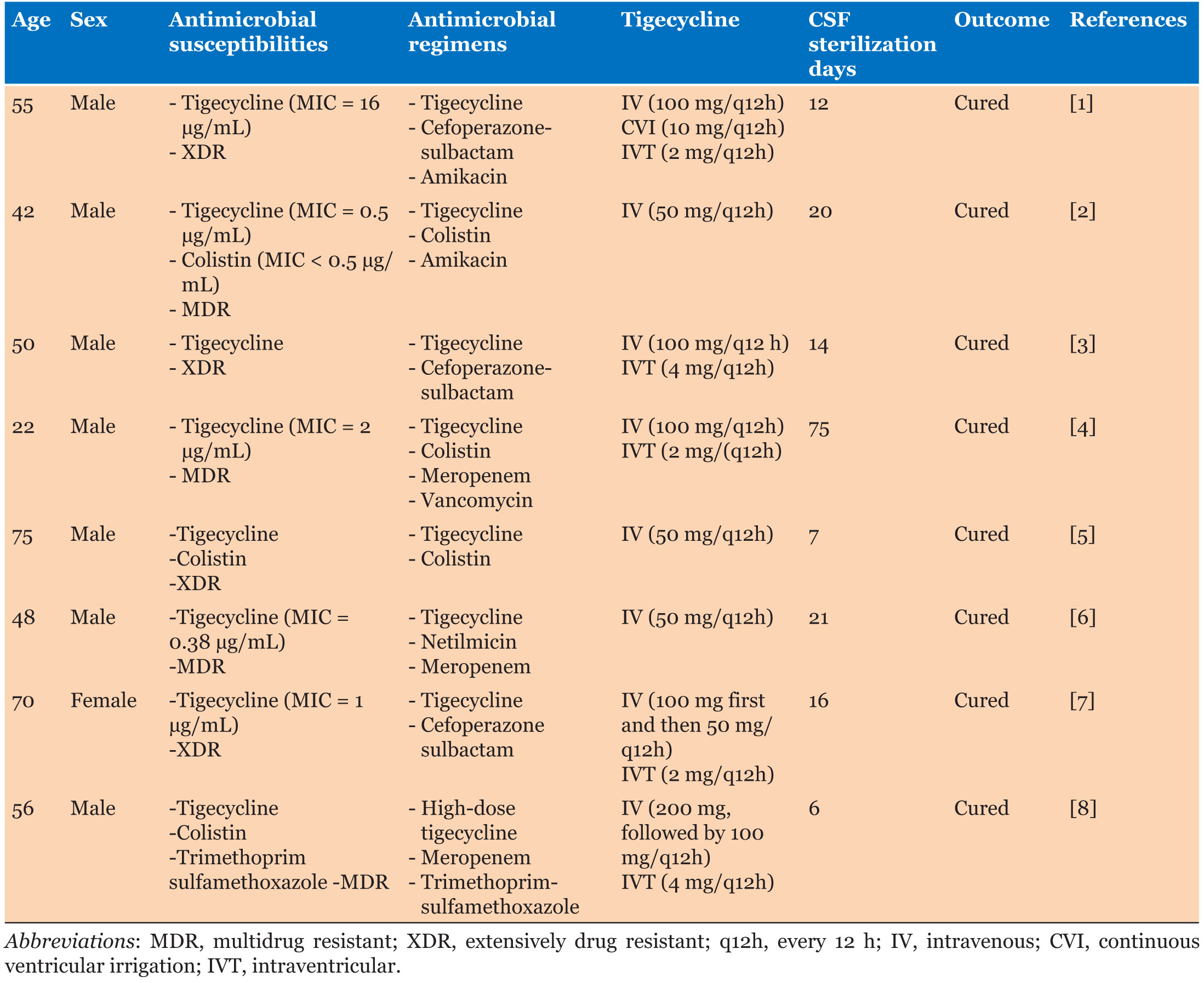
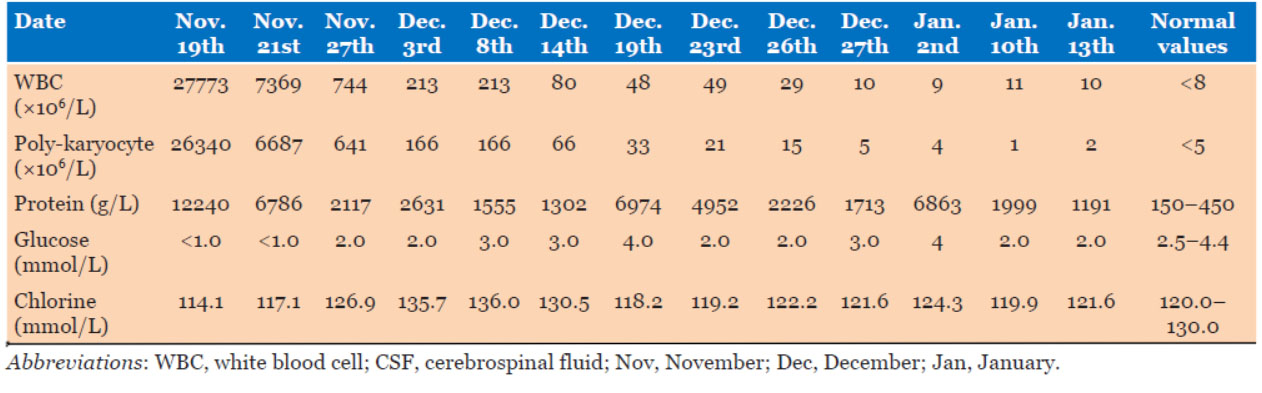
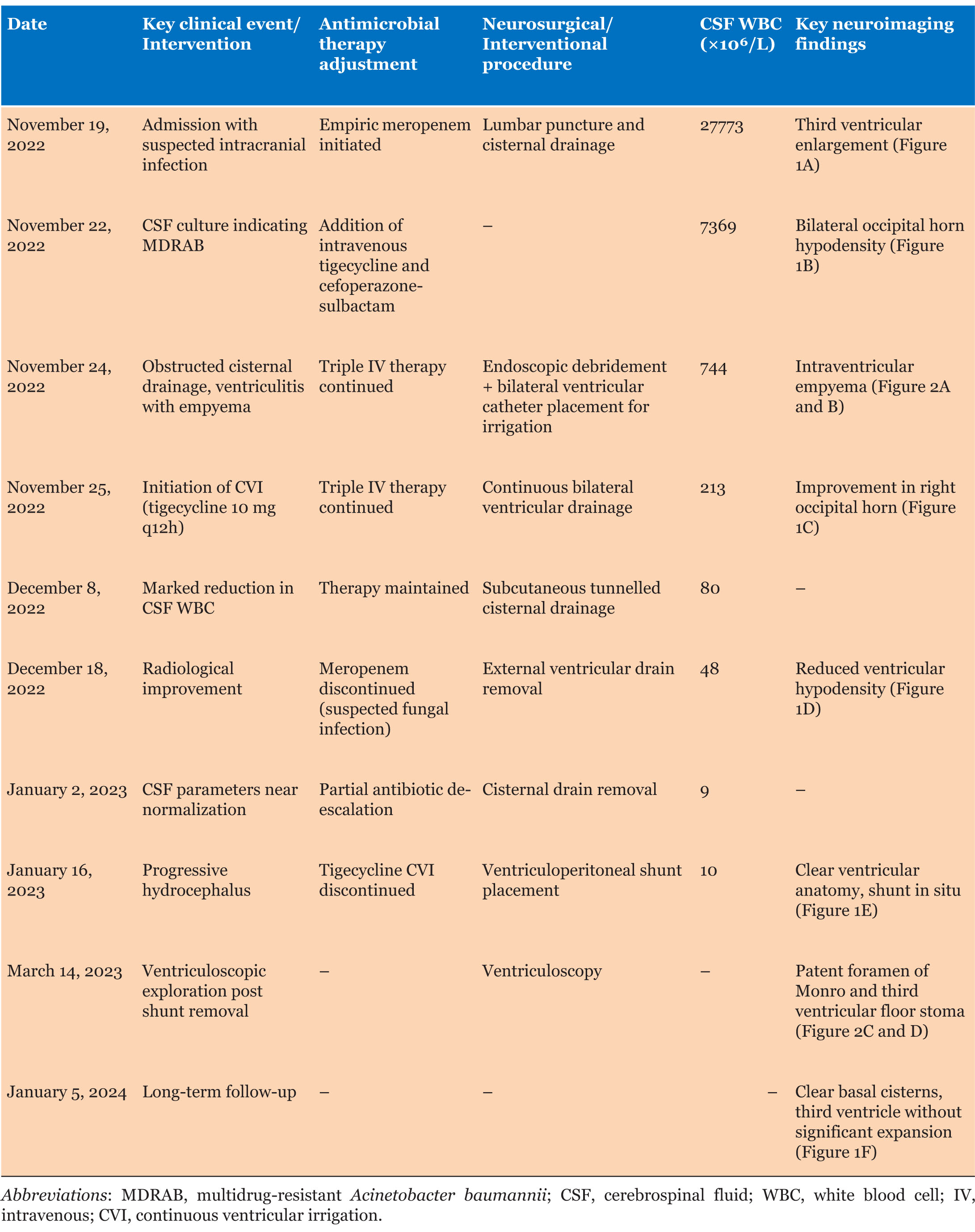
On November 19, 2022, a 50-year-old female patient was transferred to the Neurosurgery Intensive Care Unit at Huazhong University of Science and Technology Union Shenzhen Hospital. On October 27, 2022, the patient suddenly experienced impaired consciousness without apparent cause. The patient was diagnosed at another hospital with “moxaynia with ventricular hemorrhage” and underwent bilateral lateral ventriculostomy with external drainage. Twenty days after the drainage tubes were removed, the patient developed right lateral ventricle cerebrospinal fluid (CSF) leakage accompanied by fever. A follow-up cranial computed tomography (CT) scan revealed hydrocephalus. On the 22nd postoperative day, the patient was transferred to our hospital with a Glasgow coma scale (GCS) score of 5 (E1V1M3). A cranial CT performed at admission showed an enlarged third ventricle (Figure 1A). After an intracranial infection was suspected, a lumbar puncture was performed and a lumbar cisternal catheter was placed. Analysis of the retained CSF revealed a white blood cell (WBC) count of 27,773.0 × 106/L, a polymorphonuclear cell count of 26,340 × 106/L, a total microprotein concentration of 12,240 mg/L, a glucose concentration of <1.0 mmol/L, and a chlorine concentration of 114.1 mmol/L. On the basis of these findings, we initiated empirical antimicrobial therapy with meropenem (2 g every 8 hours [q8h]). On the third day after admission, a repeat cranial CT revealed effusions of the bilateral occipital horns and third ventricle with fluid-level low-density shadows (Figure 1B). On day 4, cerebrospinal fluid culture results indicated that the AB strain was susceptible only to polymyxin and tigecycline. Chest CT demonstrated pulmonary infiltrates suggestive of pneumonia, and the same AB strain was isolated from sputum.
Adjustment and Decision-Making in Antimicrobial Therapy
Given that polymyxin is currently not officially marketed in mainland China and cannot be obtained through specialized pharmacy channels within the critical clinical timeframe, we added tigecycline on the same day following multidisciplinary discussion and after obtaining informed consent from the patient’s family. Given that the efficacy of standard-dose tigecycline in severe infections remains controversial, and to achieve better antimicrobial efficacy, we adopted a high-dose tigecycline regimen. For MDRAB infections, we adopted a combination therapy strategy. The intravenous antibiotic regimen was adjusted to triple therapy with tigecycline, cefoperazone/sulbactam, and meropenem. Cefoperazone/sulbactam (2 g every 6 hours; total daily dose, 8 g) was administered off-label, with informed consent obtained from the patient’s family prior to use. The intravenous tigecycline regimen consisted of a loading dose of 200 mg followed by a maintenance dosage of 100 mg every 12 hours.
Intervention with Neurosurgery and Intraventricular Drug Administration
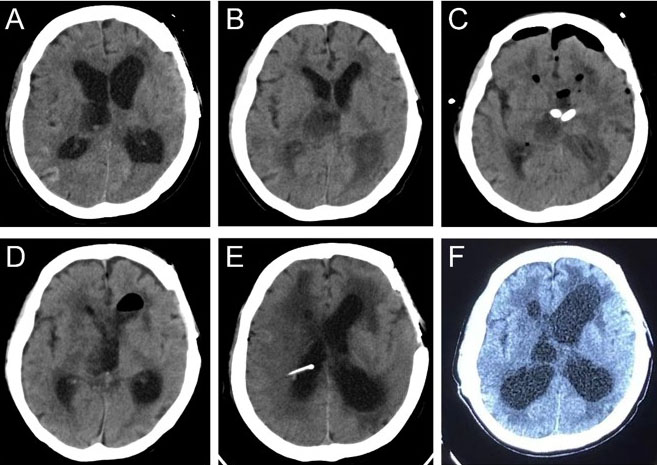
On November 24, 2022, the patient presented with obstruction of the lumbar drainage catheter and impaired drainage due to purulent cerebrospinal fluid. Following departmental consultation, a diagnosis of ventriculomyelitis was considered. A ventriculoscopic evacuation of purulent secretions and bilateral lateral ventriculostomy with irrigation were performed, during which substantial amounts of the purulent fluid were removed (Figure 2A and Figure 2B). Simultaneously, microballoon dilation was used to open the stenosed foramen of Monro, promoting CSF flow and alleviating hydrocephalus. Partial clearance of adherent purulent deposits in the ventricular horns was achieved intraoperatively. Given the limited ability of tigecycline to penetrate the blood-brain barrier, intraventricular administration was considered off-label use. After fully informing the family of risks and obtaining signed informed consent, we initiated CVI administration via the lateral ventricle drainage catheter on November 25: 10 mg of tigecycline dissolved in 100 mL of 0.9% sodium chloride solution was administered twice daily. An intravenous triple therapy regimen (100 mg q12h of tigecycline, 2 g q6h of sulbactam, and 2 g q8h of meropenem) and bilateral continuous ventricular drainage were maintained.
On the first day after lavage, follow-up cranial CT revealed persistent bilateral occipital angle fluid-filled hypodensity, although the right occipital angle improved compared with that on previous imaging (Figure 1C). Starting on postoperative day 3, multiple cerebrospinal fluid analyses demonstrated a declining trend in WBC count. On December 7, 2022, a CSF review indicated an elevated WBC count compared with previous results, potentially related to worsening drug-resistant infection or detachment of adherent pus. High-throughput sequencing of the lavage fluid revealed a cycle threshold (Ct) of 31.07, and AB was detected, with no other bacterial or fungal infections identified. On December 8, 2022, follow-up CSF analysis revealed a marked decrease in the WBC count, with CSF biochemical parameters (glucose and chloride) largely returning to normal. This finding confirmed that the previous WBC elevation was likely related to detachment of the adherent pus plaque. To establish continuous drainage between the ventricles and the lumbar cistern, the pus-like deposits were thoroughly cleared, and to prepare for gradual removal of the ventricular drain, we performed a subcutaneous tunnel drainage procedure with a lumbar cistern catheter placement on December 12, 2022. On December 14, 2022, the lateral ventricle external drainage tube was removed. High-throughput sequencing of the lumbar cistern drainage fluid revealed a Ct value of 34.72. A December 18 cranial CT scan revealed a marked reduction in fluid-filled hypodense areas in both the occipital horns and the third ventricle (Figure 1D).
On January 2, 2023 (44th day of hospitalization), a cerebrospinal fluid analysis revealed a WBC count of 9.0 × 106/L, a polymorphonuclear cell concentration of 4 × 106/L, a total protein concentration of 6863.0 mg/L, a glucose concentration of 4.0 mmol/L, and a chloride concentration of 124.3 mmol/L (Table2). The lumbar drainage tube was subsequently removed, and treatment with the same dose of tigecycline was continued. A follow-up cranial CT scan subsequently indicated worsening hydrocephalus, leading to a ventriculoperitoneal shunt procedure on January 16, 2023. Postoperative cranial CT demonstrated clear visualization of bilateral occipital horns and third ventricle structures, with the ventricular end of the shunt correctly positioned (Figure 1E). To reduce the risk of recurrent intracranial infection, tigecycline was discontinued one week after shunt placement. The total duration of antimicrobial therapy was as follows: meropenem for 37 days, intravenous tigecycline for 62 days, cefoperazone/sulbactam for 47 days, and tigecycline CVI for 19 days. On the 71st day of hospitalization, follow-up cranial CT showed mild ventricular enlargement with marked improvement in bilateral ventriculoperitoneal abscesses.
Monitoring and Management of Adverse Reactions
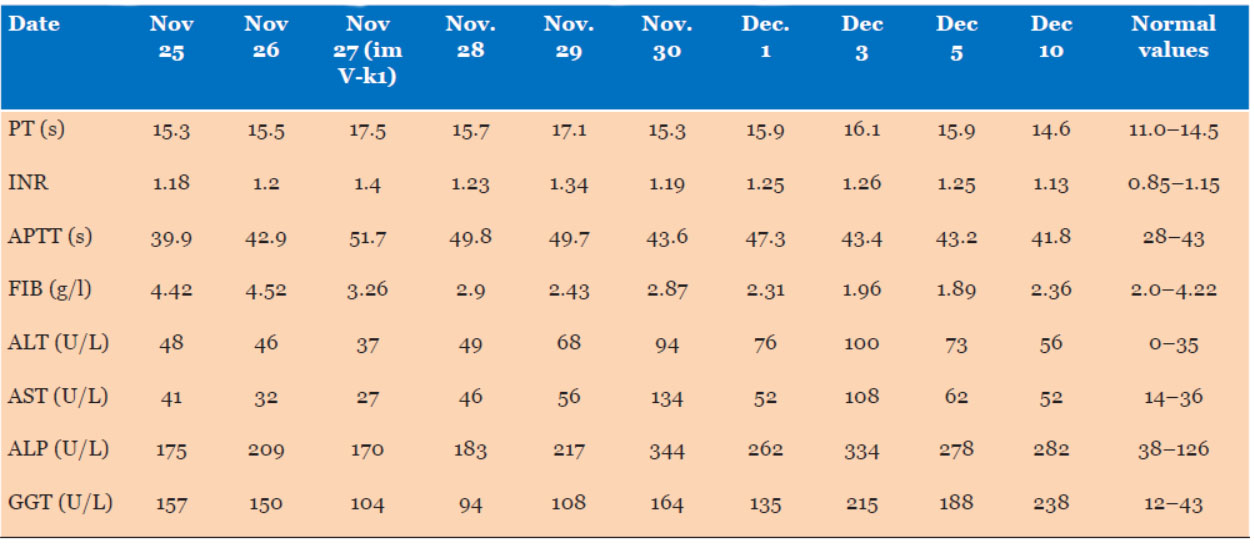
Tigecycline therapy was initiated on November 25, one day after the patient’s surgery on the 24th. Dynamic monitoring of coagulation and hepatic function markers revealed the following changes: on the 3rd day of treatment (November 27), early coagulation disturbances were noted: the prothrombin time (PT) increased to 17.5 seconds (reference range 11.0–14.5 seconds), the international normalized ratio (INR) increased to 1.4 (normal 0.85–1.15), and the fibrinogen (FIB) concentration decreased to 3.26 g/L (normal 2.0–4.22 g/L), which is consistent with mild hypocoagulability. During therapy, the patient also developed evidence of liver dysfunction. By November 30 (6th day of treatment), the alanine aminotransferase (ALT) concentration had increased to 68 U/L (normal 0–35 U/L), and the aspartate aminotransferase (AST) concentration had increased to 56 U/L (normal 14–36 U/L). On December 1 (7th day of treatment), these abnormalities worsened: ALT further increased to 94 U/L, AST to 134 U/L, alkaline phosphatase (ALP) increased to 344 U/L (normal 38–126 U/L), and gamma-glutamyl transferase (GGT) increased to 164 U/L (normal 12–43 U/L). Given the temporal alignment between the start of tigecycline and the onset of hepatic function changes, tigecycline-associated hepatotoxicity remains a plausible etiology. Targeted interventions were initiated, and the patient was placed under close clinical surveillance. A timeline summarizing the temporal changes in coagulation function and hepatic function during tigecycline therapy is presented in Table 4.
Additionally, on December 12, 2022 (23 days after meropenem initiation), the patient tested positive for (1-3)-β-D-glucan in the blood. Subsequent monitoring revealed a persistent increase in this marker, suggesting potential invasive fungal infection. A chest CT scan revealed pulmonary nodules. Following consultation with the respiratory department, these findings were considered related to prolonged meropenem use, leading to discontinuation of the drug on December 26, 2022. A timeline summarizing key clinical events, antimicrobial adjustments, surgical interventions, and laboratory/imaging changes during this case is presented in Table 3.
Treatment Outcomes and Follow-up
The patient was discharged on the 75th day of hospitalization and was in an improved condition, with a GCS score of 7 (E1V1M5); no signs of intracranial infection were observed. The dynamic changes in cerebrospinal fluid parameters during treatment are detailed in Table 4. Approximately one month after discharge, the patient developed fever, leading to the removal of the ventriculoperitoneal shunt. On March 14, 2023, endoscopic ventriculostomy revealed unobstructed communication between the interventricular foramen and the floor of the third ventricle, with no significant turbidity or flocculent material observed (Figure 2C and Figure 2D). On January 5, 2024, approximately one year after discharge, follow-up cranial CT demonstrated clear cisternal structures and no significant enlargement of the third ventricle (Figure 1F).
Discussion
With the increasing rate of antimicrobial resistance and difficulty penetrating the blood-brain barrier, the key to treating intracranial infections lies in whether drugs can cross the blood-brain barrier and achieve effective concentrations in the cerebrospinal fluid. In the absence of access to polymyxin domestically, intrathecal administration of tigecycline (off-label use) may be considered. Intraventricular administration allows the drug to directly enter the cerebrospinal fluid and diffuse toward the brain surface, achieving effective local therapeutic concentrations and thereby enhancing antimicrobial efficacy.
Ventricular meningitis caused by MDRAB infections frequently leads to empyema and adhesions, posing significant treatment challenges. Neuroendoscopy represents an effective approach for treating purulent ventricular meningitis, offering the advantage of safely removing inflammatory material from ventricular walls under direct visualization while completely draining infected CSF. If empyema has developed, bilateral ventricular lavage can sustain drainage of the purulent fluid. In combination with systemic antibiotic therapy, intraventricular antibiotic lavage enhances local drug efficacy by clearing pus, reducing adhesions, and improving CSF circulation, thereby effectively controlling infection. Combined with continuous external ventricular drainage, it promotes CSF flow and effectively prevents hydrocephalus.
The rationale for the triple-agent intravenous regimen of meropenem, cefoperazone-sulbactam, and tigecycline in this case is as follows: meropenem and cefoperazone/sulbactam both exhibit activity against AB, and their combination may produce synergistic effects while delaying the emergence of resistance. Regarding the use of tigecycline, the high-dose regimen employed in this case (a loading dose of 200 mg followed by 100 mg twice daily) was based on its pharmacokinetic properties and existing clinical evidence. Previous studies have shown that standard-dose tigecycline (a loading dose of 100 mg, followed by 50 mg twice daily) is no more effective than other antimicrobial agents in treating severe infections and may even be associated with higher mortality rates [9],[10],[11]. Pharmacokinetic and pharmacodynamic research has indicated that the inability to achieve sufficient plasma concentrations at standard doses may be the primary cause of suboptimal efficacy [12]. Consequently, highdose regimens are adopted in some clinical settings for severe infections caused by multidrug-resistant bacteria. A meta-analysis involving 593 patients confirmed that, compared with standard-dose treatment, high-dose tigecycline reduces overall mortality and improves clinical cure rates and microbial eradication rates [13]. This evidence supports the rationale for our strategy of combining high-dose intravenous tigecycline with local therapy in the treatment of MDRAB ventriculitis in this case. Although tigecycline has limited ability to cross the blood-brain barrier, high-dose systemic administration combined with intraventricular irrigation played a critical role in controlling systemic infection and augmenting local treatment. This combination regimen has been reported in select previous cases of MDRAB intracranial infection, although its synergistic effects and cumulative toxicity require further research.
Compared with other reported MDRAB meningitis/ventriculitis cases (Table 1 in the original text), this case shares similarities in terms of the use of a combination of intravenous and intraventricular administration strategies. However, its distinctiveness lies in combining CVI with neuroendoscopic debridement, making it more suitable for cases complicated by ventriculomycosis. Additionally, the prolonged duration of antimicrobial therapy in this case may be related to the severity of infection and associated complications.
The observed coagulation dysfunction and elevated liver enzymes align with known adverse reactions to tigecycline, indicating the need for close monitoring of coagulation and liver function during prolonged or high-dose therapy. Furthermore, although polymyxin is the first-line treatment for CNS infections caused by MDRAB, as recommended by international guidelines, its unavailability in this region influenced treatment decisions. This underscores the necessity of developing individualized treatment strategies for drug-resistant infections on the basis of drug availability, patient-specific circumstances, and antimicrobial susceptibility testing results.
Conclusion
This case demonstrates that continuous ventricular lavage combined with intravenous tigecycline has potential for treating MDRAB ventriculitis, particularly when polymyxin is unavailable and blood–brain barrier penetration is limited. However, as this is a single case report, its efficacy and safety require validation through larger-scale clinical studies. When intraventricular antibiotics are used off-label, risks should be thoroughly assessed. Individualized treatment plans should be developed in conjunction with neurosurgical intervention and multidisciplinary collaboration.
REFERENCES
1.
Long W, Yuan J, Liu J, Liu J, Wu M, Chen X, et al. Multidrug resistant brain abscess due to Acinetobacter baumannii ventriculitis cleared by intraventricular and intravenous tigecycline therapy: A case report and review of literature. Front Neurol 2018;9:518. [CrossRef]
[Pubmed]

2.
De Pascale G, Pompucci A, Maviglia R, Spanu T, Bello G, Mangiola A, et al. Successful treatment of multidrug-resistant Acinetobacter baumannii ventriculitis with intrathecal and intravenous colistin. Minerva Anestesiol 2010;76(11):957–60.
[Pubmed]

3.
Fang YQ, Zhan RC, Jia W, Zhang BQ, Wang JJ. A case report of intraventricular tigecycline therapy for intracranial infection with extremely drug resistant Acinetobacter baumannii. Medicine (Baltimore) 2017;96(31):e7703. [CrossRef]
[Pubmed]

4.
Lauretti L, D’Alessandris QG, Fantoni M, D’Inzeo T, Fernandez E, Pallini R, et al. First reported case of intraventricular tigecycline for meningitis from extremely drug-resistant Acinetobacter baumannii. J Neurosurg 2017;127(2):370–3. [CrossRef]
[Pubmed]

5.
Shrestha GS, Tamang S, Paneru HR, Shrestha PS, Keyal N, Acharya SP, et al. Colistin and tigecycline for management of external ventricular device-related ventriculitis due to multidrug-resistant Acinetobacter baumannii. J Neurosci Rural Pract 2016;7(3):450–2. [CrossRef]
[Pubmed]

6.
Tutuncu EE, Kuscu F, Gurbuz Y, Ozturk B, Haykir A, Sencan I. Tigecycline use in two cases with multidrug-resistant Acinetobacter baumannii meningitis. Int J Infect Dis 2010;14 Suppl 3:e224–6. [CrossRef]
[Pubmed]

7.
Liu Y, Pu Z, Zhao M. Case report of successful treatment of extensively drug-resistant acinetobacter Baumannii ventriculitis with intravenous plus intraventricular tigecycline. Antimicrob Agents Chemother 2018;62(11):e01625–18. [CrossRef]
[Pubmed]

8.
Abdallah M, Alsaleh H, Baradwan A, Alfawares R, Alobaid A, Rasheed A, et al. Intraventricular tigecycline as a last resort therapy in a patient with difficult-to-treat healthcare-associated Acinetobacter baumannii ventriculitis: A case report. SN Compr Clin Med 2020;2(9):1683–7. [CrossRef]
[Pubmed]

9.
Tasina E, Haidich AB, Kokkali S, Arvanitidou M. Efficacy and safety of tigecycline for the treatment of infectious diseases: A meta-analysis. Lancet Infect Dis 2011;11(11):834–44. [CrossRef]
[Pubmed]

10.
Shen F, Han Q, Xie D, Fang M, Zeng H, Deng Y. Efficacy and safety of tigecycline for the treatment of severe infectious diseases: An updated meta-analysis of RCTs. Int J Infect Dis 2015;39:25–33. [CrossRef]
[Pubmed]

11.
Prasad P, Sun J, Danner RL, Natanson C. Excess deaths associated with tigecycline after approval based on noninferiority trials. Clin Infect Dis 2012;54(12):1699–709. [CrossRef]
[Pubmed]

12.
Giamarellou H, Poulakou G. Pharmacokinetic and pharmacodynamic evaluation of tigecycline. Expert Opin Drug Metab Toxicol 2011;7(11):1459–70. [CrossRef]
[Pubmed]

13.
Zha L, Pan L, Guo J, French N, Villanueva EV, Tefsen B. Effectiveness and safety of high dose tigecycline for the treatment of severe infections: A systematic review and meta-analysis. Adv Ther 2020;37(3):1049–64. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Yanting Chen - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Yi Feng - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Xuemei Huang - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guoqiang Qin - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2026 Yanting Chen et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.