|
|
Case Report
| ||||||
| Neurocysticercosis: A case report | ||||||
| Smit Shah1, Rachana Tyagi2 | ||||||
|
1Rutgers, Robert Wood Johnson Medical School, Medical Student, 675 Hoes Lane West, Piscataway, New Jersey 2Navicent Health, Director of Pediatric Neurosurgery, Macon, Georgia, 777 Hemlock St, Macon, Georgia | ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article] [Similar article in PubMed] [Similar article in Google Scholar] |
| How to cite this article |
| Shah S, Tyagi R. Neurocysticercosis: A case report. J Case Rep Images Infect Dis 2018;1:100001Z16SS2018. |
|
ABSTRACT
| ||||||
|
Introduction: Parasitic diseases are a growing global public health concern with severe neurological consequences. It is a leading cause of epilepsy in developing countries including India, Africa, Latin America and China. Case Report: In this paper we discuss a case of a 34-year-old female patient with a past medical history of unsuccessfully repaired ano-urethral fistula who was diagnosed with Neurocysticercosis (NCC) after initially presenting with seizures. In addition, we also discuss how this patient was non-surgically managed after radiological and serological confirmation of NCC. Besides case details, we also elaborate on the parasitology of the tapeworm Taenia Solium along with potential sources of infection in this case. Finally, we also discuss, different ways of classifying NCC based on literature review. Conclusion:Thus, this case report highlights a classic presentation of NCC due to inappropriate personal hygiene along with image findings and clinical management. Keywords: Albendazole, Fecal-Oral transmission, Neurocysticercosis, Seizures | ||||||
|
INTRODUCTION
| ||||||
|
Neurocysticercosis is a parasitic infection caused by the tapeworm Taenia Solium. It is a leading cause of epilepsy in developing countries including India, Africa, Latin America and China [1]. Staging of cysticerci can be characterized by MRI: Vesicular cysts are stage one and colloidal cysts are stage two [2]. These cysts can progress to stage three which has granular nodular degeneration with onset of calcification and finally stage four, or complete calcification on CT and MRI [2]. Most commonly patients present with new onset partial seizures with or without secondary generalization due to cyst formation in various areas of the brain parenchyma. The two most common types of cysts are (1) Vesicular cysts which are less epileptogenic and have less mass effect on imaging and (2) Colloid cyst which consist of gelatinous material that exhibits ring enhancement and edema on imaging which is associated with increased epileptogenic potential [1]. Alternatively, patients may present with a generalized headache secondary to increased intracranial pressure and meningitis. Hallmarks of NCC include variation in clinical manifestations as well as disease severity that can vary from completely asymptomatic infection to severe disease and death [3]. In addition, the number, size, location and intensity of cysts also lead to varied symptomatic presentations in patients [3]. Generally, cysts in brain parenchyma that present as new-onset seizures can be effectively controlled initially by antiepileptics along with antihelminthics after confirmation of diagnosis. In addition, many cases of NCC have been linked to primary medical conditions like urticarial vasculitis [4]. The health care costs attributed to NCC also pose a major problem, particularly as the indigent population has a greater disease burden than patients in a higher socioeconomic group. In a recent study conducted in Mexico, NCC patients required intensive treatment (hospitalization and/or surgical intervention for diagnosis) leading to huge amount of heath care costs [5]. | ||||||
|
CASE REPORT
| ||||||
|
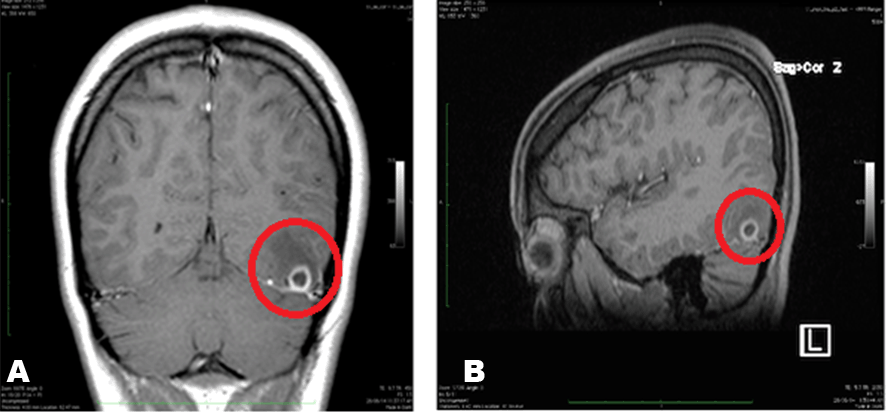
Our patient is a 34-year-old female with a past medical history of type 2 Diabetes Mellitus (BMI 31 kg/m2) who was brought to the emergency room with a new onset tonic clonic seizure witnessed by her brother. Upon arrival, patient was postictal and drowsy. On physical examination, she was awake, responsive but confused with a right sided tongue bite without any focal neurological deficits. Initial treatment with 100% oxygen and intramuscular phenobarbital was administered and an urgent CT scan was ordered along with neurology consult. CT Scan was negative. Patient and her relatives denied any history of seizures. However, patient did have a congenital ano-urethral fistula which was unsuccessfully repaired in the late 1980s after which a colostomy bag was placed in right lower quadrant of her abdomen. In order to ensure good hygiene, patient used warm water enemas to clean the colostomy site daily. Patient worked as an interior designer. No smoking, drinking or illicit drug use as per patient's relatives. On further interview, patient's brother reported that she made a recent dietary change by switching from being vegan to a non-vegan diet which included prepacked salads with meat and pork 6 months prior to admission. Due to a higher possibility of an infectious etiology, MRI of brain was ordered. MRI demonstrated a well-defined ring enhancing cystic lesion in subcortical basal occipital lobe with eccentric poorly enhancing calcified scolex with perilesional edema suggesting granulomatous NCC as shown in Figure 1(A and B) . Blood tests revealed elevated IgE levels along with (+) CSF ELISA testing. Awake EEG was performed with interictal bilateral frontal temporal spike wave epileptiform discharges although no discharges were noted during photic stimulation with uneventful sleep recordings as well. Stool culture and analysis was positive for adult tapeworms. Consequently, patient was started on Phenytoin, then switched to Valproic Acid for seizure control. In addition, patient was started on Albendazole for one year with initial follow up every two weeks for first six months. During initial follow up patient had no recurrent seizures. In addition, patient has been seizure free for more than one year after initial treatment. In addition, patient has regularly followed up with good medication compliance, improved personal hygiene and returning to a vegetarian diet. | ||||||
|
DISCUSSION
| ||||||
|
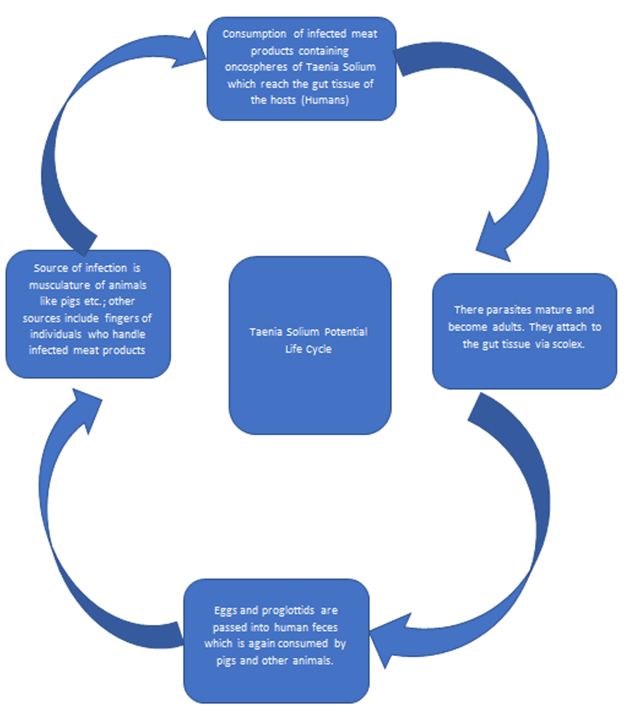
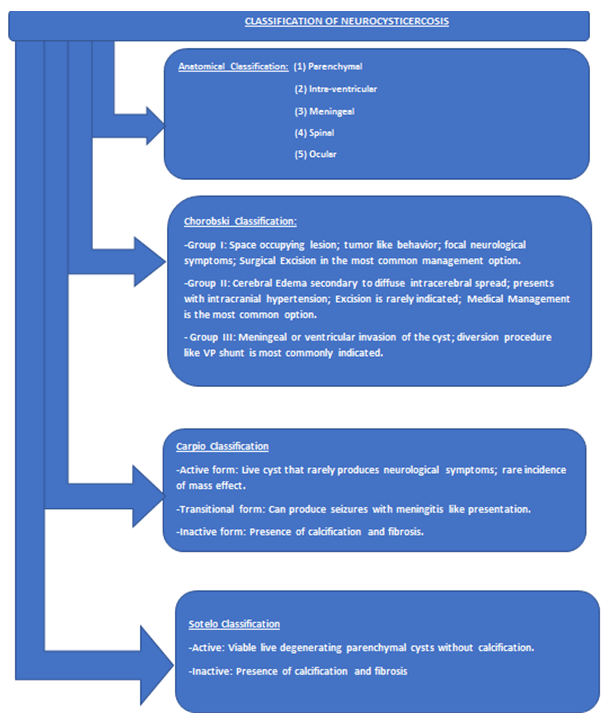
Based on this case report, we believe the three possible sources of tapeworm infection are from the contamination of the pre-packaged salad by the food workers, self-infection from poor hand-washing after her enemas and direct entry of the parasite eggs through mucosal tears in the colon after enemas. This case possible presents a unique method of self-infection due to inappropriate personal hygiene i.e. lack of hand washing after warm water enemas at colostomy site. In the southwestern US and third world countries, NCC has been a major public health problem and also one of leading causes of epilepsy [1]. Croker et. al determined that there were about 300 cases of NCC in Southern California in 2009, in which men were more likely to be affected as compared to women [6]. This study demonstrated the economic toll of treatment for NCC. The cost for hospitalization was 5.1 million dollars and average length of stay was 6.5 days as compared to 4.6 days average LOS for all US admissions [6]. In terms of average cost of hospitalization, total inflation-adjusted hospitalization charges [for NCC] over the 10-year study period were $908,238,000, increasing 27% in 2003 to 2012 [7]. Cases like this, also expose the risk of spreading disease though the global food supply chain with one possible route of transmission being pork contaminated with T. Solium (Figure 2) in the imported salads [8]. Like other infectious diseases, Neurocysticercosis outbreaks financially burden countries that are economically well off as well as underdeveloped nations [9],[10]. Neurocysticercosis poses a major health hazard in close communities where people have a high likelihood of infection because of exposure to infected feces that contains tapeworm eggs. Figure 3 demonstrates the different classification of Neurocysticercosis [11], [12], [13], [14], [15]. Due to increasing global migration, NCC has a great potential for rapid spread. Since carriers may be asymptomatic, tracking the infectious source can become very difficult and expensive for health investigators. Prevention strategies must include proper food surveillance via stricter controls implemented via WHO standards and most importantly thorough history and physical examination of patients with attention to their diet & lifestyle. Public awareness and early diagnosis will allow treatment before severe neurologic complications occur, decreasing costs to the patient and society as a whole. | ||||||
| ||||||
| ||||||
| ||||||
|
CONCLUSION
| ||||||
|
Neurocysticercosis has the potential of becoming a much more prevalent disease with high societal and individual costs. The spread of disease may be decreased by careful attention to the safety of the global food supply chain and public education campaigns. | ||||||
|
REFERENCES
| ||||||
| ||||||
|
SUGGESTED READING
| ||||||
|
| ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions
Smit Shah – Substantial contributions to conception and design, Acquisition of data, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Rachana Tyagi – Substantial contributions to conception and design, Acquisition of data, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor of Submission
The corresponding author is the guarantor of submission. |
|
Source of Support
None |
|
Consent Statement
Written informed consent was obtained from the patient for publication of this study. |
|
Conflict of Interest
Author declares no conflict of interest. |
|
Copyright
© 2018 Smit Shah et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|